It helped identify a significant number of additional organisms in the study that were missed or not reported by standard of care testing alone, with an overall weighted average sensitivity of 96.9% and an overall weighted average specificity of 97.1%.” 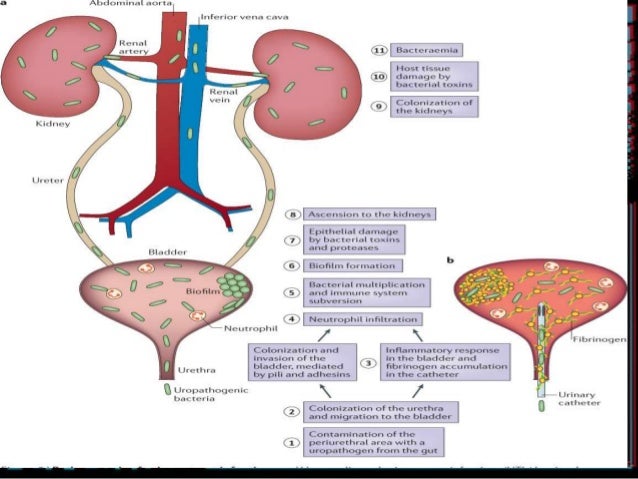
“Our data demonstrated that the Unyvero UTI panel covered nearly 94% of all organisms identified in standard of care microbiology culture. “As discussed during my presentation, analysis and evaluation of such a large clinical trial data set was a challenge, given the complexity of result quantitation, multiple reference methods and the frequent presence of flora in urine samples,” explained Dr. OpGen is seeking marketing authorization for its Unyvero UTI panel. Food and Drug Administration (FDA) was submitted in April 2023 and is currently under Substantive Review by the FDA. The company’s De Novo classification request to the U.S. The test aims at quantitative detection of microorganisms. OpGen’s Unyvero UTI panel tests for a broad range of bacterial and fungal pathogens as well as antimicrobial resistance markers directly from urine specimens. Based on over 1,800 patient samples, the presentation titled “Multi-Center Clinical Trial Using a Novel Semi-Quantitative Multiplex-PCR Based Diagnostic Panel for Urinary Tract Specimens” summarized the objectives of the clinical trial, the Unyvero UTI panel coverage and result evaluation recommendations based on multiple reference testing methods applied during the study. Matthias Klein has presented results from the clinical trial for its Unyvero Urinary Tract Infection Panel during ASM Microbe’s In Depth Symposium session ‘The Year in Clinical Microbiology’ on June 17 th in Houston, TX. (Nasdaq: OPGN, “OpGen” or “the Company”), a precision medicine company harnessing the power of molecular diagnostics and bioinformatics to help combat infectious disease, reported that its Principal Scientist Dr. ROCKVILLE, Md., J(GLOBE NEWSWIRE) - OpGen, Inc. Presentation focused on primary study endpoint and recommendations for result evaluationĭe Novo request for Unyvero Urinary Tract Infection (UTI) currently under Substantive Review by the FDAĪdditionally, data presented at the conference on Unyvero Implant and Tissue Infection (ITI) study from the University of Cincinnati ‘Multi-Center Clinical Trial Using a Novel Semi-Quantitative Multiplex-PCR Based Diagnostic Panel for Urinary Tract Specimens’ presented on June 17 th, 2023 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
